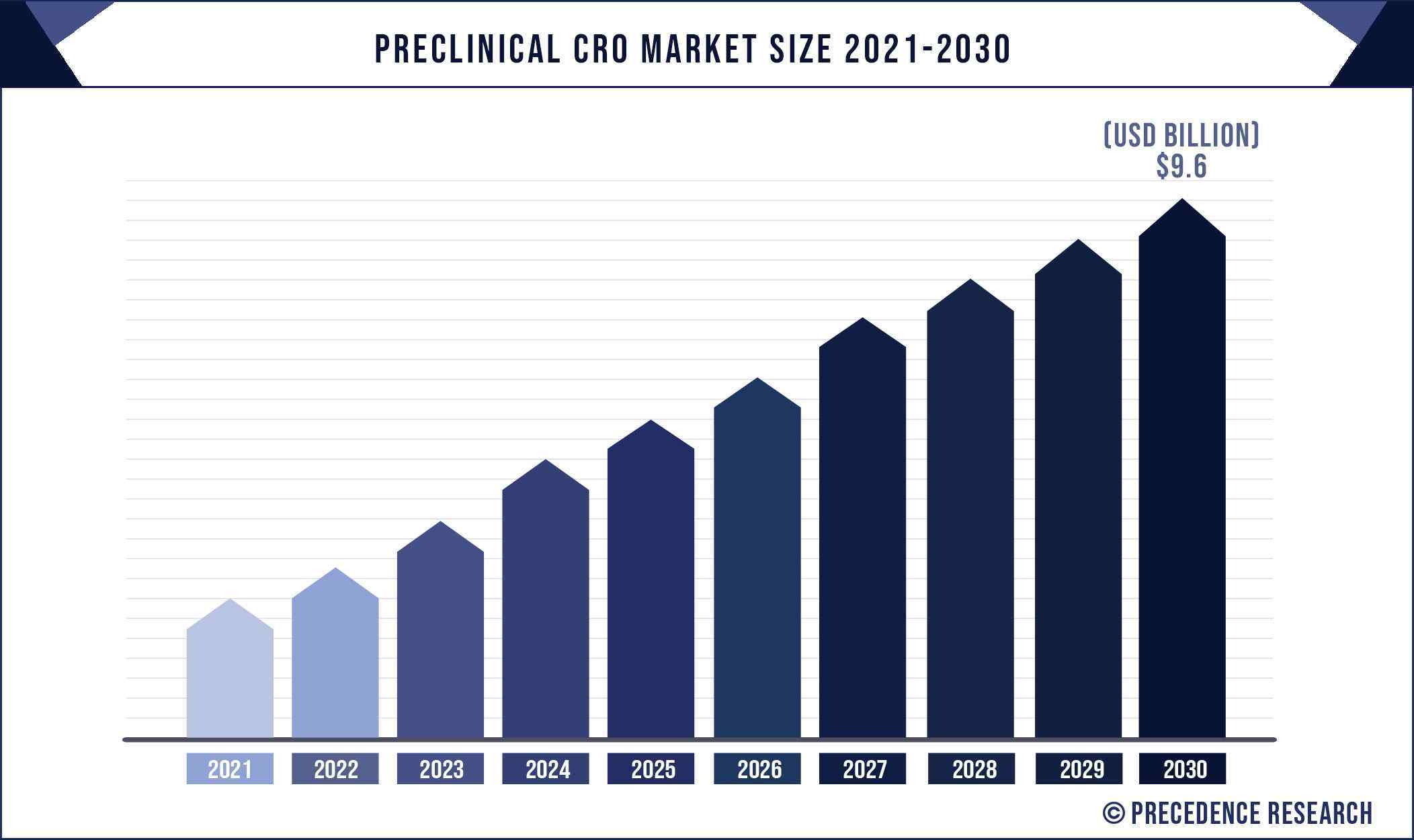
The global preclinical CRO market size is expected to reach around US$ 9.6 billion by 2030 from US$ 4.9 billion in 2020 and is expected to grow at an impressive double-digit rate of 7.5% from 2021 to 2030.
The study includes drivers and restraints of this market. The study provides an analysis of the global preclinical CRO market for the period 2017-2030, wherein 2021 to 2030 is the forecast period and 2020 is considered as the base year.
We customize your report according to your research need. REQUEST CUSTOMIZED COPY OF REPORT (Including TOC, List of Tables & Figures, and Chart) @ https://www.precedenceresearch.com/customization/1361
The global medical tourism market is primarily driven by the increased demand for the cheap treatment for various chronic diseases available in the developing countries. Sometimes, the treatment of certain diseases are quite expensive in the home country, therefore, in order to obtain cheap and advanced treatments people opt for visiting foreign countries. The rising burden of diseases among the global population and increased affordability of various latest therapies is fostering the growth of the medical tourism market. Globalization is an essential aspect of the medical tourism industry. Globalization resulted in the free flow of medical technologies, health professionals, patients, medicines, and capital funds which constitutes the development of the medical tourism industry. The rising government investments in the development of tourism infrastructure and building strong and advanced healthcare facilities are significantly propelling the growth of the global medical tourism market.
The rising prevalence of chronic diseases and availability of various treatments related to fertility, cancer, cardiovascular health, and orthopedics is augmenting the demand for the medical tourism market. The medical tourism market is dominantly affected by various factors such as climate, geo-political relations between countries, political stability, and local regulations. Favorable government policies related to VISA and other permits plays a crucial role in the growth of the market.
Based on region, Asia Pacific is the dominating and the fastest-growing market in the global tourism market. This can be attributed to the availability of cheap air transport and availability of specialized, advanced, and affordable treatments of various chronic diseases. Furthermore, the availability of cheap cosmetic surgery and cosmetic treatment facilities is boosting the growth of the medical tourism in this region. Cosmetics treatment is not covered in the healthcare policies and hence, people are increasingly preferring Asia Pacific for the availability of cheap cosmetic treatments. Further, the increasing government initiatives to promote tourism in the region coupled with the rising investments for the development of sophisticated healthcare infrastructure is expected to contribute exponentially towards the market growth in the forthcoming years. The countries such as Thailand, Singapore, India, and China are the major destinations for the medical tourists owing to the availability of advanced medical technologies at affordable prices. The rising burden of diseases in the developed regions like North America and Europe is attracting medical tourists in the Asia Pacific region.
The medial tourism market is highly dependent of the foreign policies, regulations, and geo-political relations between countries that may have a negative impact on the growth of the medical tourism market. Further, the availability of advanced treatment at higher costs in the regions like Europe and North America may hinder the market growth during the forecast period.
The rapid development of various new technologies and increasing investments in the research activities to develop improved and effective medicines and medical devices is expected to present a huge growth opportunity for the market players in the foreseeable future. For instance, the development of targeted therapy, gene-targeted therapy, laser treatment, and various other advanced treatments are the major growth drivers of the global medical treatment market.
The recent outbreak of the COVID-19 pandemic has negatively affected the medical tourism market. Almost all the countries on this planet sealed their borders and shut down their destinations for the tourists. Most of the scheduled medical tourism were postponed or delayed for an indefinite time period. Further, disruptions in the supply chain resulted in the shortages of medicines and medical devices owing to the lack of raw materials. Hence, such pandemics may have a devastating effect on the market in the forthcoming years.
Based on the type, the cancer segment dominated the global medical tourism market. This is simply attributed to the huge demand for the cancer treatment at cheaper costs. Furthermore, the prevalence of cancer is on the rise among the global population.According to the International Agency for Research on Cancer, around 19.3 million new cancer cases and around 10 million cancer related deaths were reported in the year 2020, across the globe. Prevalence of breast cancer in the female population is surging, accounting for around 11.7% of the new cancer cases followed by the lungs cancer that accounted for 11.4% and colorectal cancer accounted for 10.0% in 2020. Hence, the availability of advanced treatment for cancer is expected to drive the segment growth and is estimated to retain its dominance throughout the forecast period.
The market is moderately fragmented with the presence of several local companies. These market players are striving to gain higher market share by adopting strategies, such as investments, partnerships, and acquisitions & mergers. Companies are also spending on the development of improved products. Moreover, they are also focusing on maintaining competitive pricing.
Why should you invest in this report?
If you are aiming to enter the global preclinical CRO market, this report is a comprehensive guide that provides crystal clear insights into this niche market. All the major application areas for preclinical CRO are covered in this report and information is given on the important regions of the world where this market is likely to boom during the forecast period of 2021-2030, so that you can plan your strategies to enter this market accordingly.
Besides, through this report, you can have a complete grasp of the level of competition you will be facing in this hugely competitive market and if you are an established player in this market already, this report will help you gauge the strategies that your competitors have adopted to stay as market leaders in this market. For new entrants to this market, the voluminous data provided in this report is invaluable.
Some of the prominent players in the global preclinical CRO market include:
- Wuxi AppTec
- Pharmaceutical Product Development
- Medpace, Inc.
- Charles River Laboratories International, Inc.
- PRA Health Science, Inc.
- PAREXEL
- Envigo
- Eurofins Scientific.
- Laboratory Corporation of America
- ICON Plc
Market Segmentation:
By Service
- Toxicology Testing
- Bioanalysis & DMPK Studies
- Chemistry
- Compound Management
- Safety Pharmacology
- Others
By End Use
- Medical Device Companies
- Biopharmaceutical
- Government & Academic Institutes
Regional Analysis:
The geographical analysis of the global preclinical CRO market has been done for North America, Europe, Asia-Pacific, and the Rest of the World.
The North American Market is again segmented into the US, Canada, and Mexico. Coming to the European Market, it can be segmented further into the UK, Germany, France, Italy, Spain, and the rest. Coming to the Asia-Pacific, the global preclinical CRO Market is segmented into China, India, Japan, and Rest of Asia Pacific. Among others, the market is segmented into the Middle East and Africa, (GCC, North Africa, South Africa and Rest of the Middle East & Africa).
Key Questions Answered by the Report:
- What will be the size of the global preclinical CRO market in 2030?
- What is the expected CAGR for the preclinical CRO market between 2021 and 2030?
- Which are the top players active in this global market?
- What are the key drivers of this global market?
- How will the market situation change in the coming years?
- Which region held the highest market share in this global market?
- What are the common business tactics adopted by players?
- What is the growth outlook of the global preclinical CRO market?
TABLE OF CONTENT
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Preclinical CRO Market
5.1. COVID-19 Landscape: Preclinical CRO Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Preclinical CRO Market, By Service
8.1. Preclinical CRO Market, by Service Type, 2021-2030
8.1.1. Toxicology Testing
8.1.1.1. Market Revenue and Forecast (2019-2030)
8.1.2. Bioanalysis & DMPK Studies
8.1.2.1. Market Revenue and Forecast (2019-2030)
8.1.3. Chemistry
8.1.3.1. Market Revenue and Forecast (2019-2030)
8.1.4. Compound Management
8.1.4.1. Market Revenue and Forecast (2019-2030)
8.1.5. Safety Pharmacology
8.1.5.1. Market Revenue and Forecast (2019-2030)
8.1.6. Others
8.1.6.1. Market Revenue and Forecast (2019-2030)
Chapter 9. Global Preclinical CRO Market, By End Use
9.1. Preclinical CRO Market, by End Use, 2021-2030
9.1.1. Medical Device Companies
9.1.1.1. Market Revenue and Forecast (2019-2030)
9.1.2. Biopharmaceutical
9.1.2.1. Market Revenue and Forecast (2019-2030)
9.1.3. Government & Academic Institutes
9.1.3.1. Market Revenue and Forecast (2019-2030)
Chapter 10. Global Preclinical CRO Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Service (2019-2030)
10.1.2. Market Revenue and Forecast, by End Use (2019-2030)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Service (2019-2030)
10.1.3.2. Market Revenue and Forecast, by End Use (2019-2030)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Service (2019-2030)
10.1.4.2. Market Revenue and Forecast, by End Use (2019-2030)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Service (2019-2030)
10.2.2. Market Revenue and Forecast, by End Use (2019-2030)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Service (2019-2030)
10.2.3.2. Market Revenue and Forecast, by End Use (2019-2030)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Service (2019-2030)
10.2.4.2. Market Revenue and Forecast, by End Use (2019-2030)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Service (2019-2030)
10.2.5.2. Market Revenue and Forecast, by End Use (2019-2030)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Service (2019-2030)
10.2.6.2. Market Revenue and Forecast, by End Use (2019-2030)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Service (2019-2030)
10.3.2. Market Revenue and Forecast, by End Use (2019-2030)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Service (2019-2030)
10.3.3.2. Market Revenue and Forecast, by End Use (2019-2030)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Service (2019-2030)
10.3.4.2. Market Revenue and Forecast, by End Use (2019-2030)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Service (2019-2030)
10.3.5.2. Market Revenue and Forecast, by End Use (2019-2030)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Service (2019-2030)
10.3.6.2. Market Revenue and Forecast, by End Use (2019-2030)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Service (2019-2030)
10.4.2. Market Revenue and Forecast, by End Use (2019-2030)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Service (2019-2030)
10.4.3.2. Market Revenue and Forecast, by End Use (2019-2030)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Service (2019-2030)
10.4.4.2. Market Revenue and Forecast, by End Use (2019-2030)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Service (2019-2030)
10.4.5.2. Market Revenue and Forecast, by End Use (2019-2030)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Service (2019-2030)
10.4.6.2. Market Revenue and Forecast, by End Use (2019-2030)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Service (2019-2030)
10.5.2. Market Revenue and Forecast, by End Use (2019-2030)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Service (2019-2030)
10.5.3.2. Market Revenue and Forecast, by End Use (2019-2030)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Service (2019-2030)
10.5.4.2. Market Revenue and Forecast, by End Use (2019-2030)
Chapter 11. Company Profiles
11.1. Wuxi AppTec
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Pharmaceutical Product Development
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. Medpace, Inc.
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. Charles River Laboratories International, Inc.
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. Recent Initiatives
11.5. PRA Health Science, Inc.
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. PAREXEL
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. Envigo
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
11.8. Eurofins Scientific.
11.8.1. Company Overview
11.8.2. Product Offerings
11.8.3. Financial Performance
11.8.4. Recent Initiatives
11.9. Laboratory Corporation of America
11.9.1. Company Overview
11.9.2. Product Offerings
11.9.3. Financial Performance
11.9.4. Recent Initiatives
11.10. ICON Plc
11.10.1. Company Overview
11.10.2. Product Offerings
11.10.3. Financial Performance
11.10.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
Buy Full Research Report (Single User License US$ 4500) @ https://www.precedenceresearch.com/checkout/1361
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com



0 Comments